“Yah, yah, yah, yah…,” yelled Zena Warrior Princess as she blinded everyone on the battle field with her bright shining teeth. Remember that old TV show where Lucy Lawless played Zena Warrior princess? Remember how white her teeth were? Wow, I did. How about Erik Estrada on Chips? Or Callie on Grey’s Anatomy? It seems everyone wants to have the brightest and whitest teeth possible. Even to a point that they look completely unnatural. Chicklets. White refrigerator doors.
The modern tooth whitening process started back in the late 1980’s when a general dentist named John Munro, was looking for a better post-scaling/root planing dressing and medication. He would make pre-operative suck down trays of the patient’s mouth and put into it a special preparation he made of 10% carbamide peroxide. I’m not really sure if it helped with the patient’s post-operative healing (cold sensitivity), but it sure did whiten their teeth! In 1989, Omni International (now part of 3M) came out with the first commercially available tooth whitening product called White & Brite. Omni started doing research with the University of Alabama on this product line, and charged doctors an up front $895 fee just to have the rights to purchase the whiten agent in the first place. Then each kit cost an arm and a leg to purchase after that. I have to admit it was worth it at the time. People flocked to get their teeth whitened. I whitened my teeth and others in my family looked like they had not brushed their teeth in weeks/months/years compared to mine. It was the beginning of a teeth whitening revolution!
Now the market is literally flooded with tooth whitening products. Some are pretty good and others are not. The use of lasers and ultraviolet lights are currently being used in some dental offices and the jury is still out on their effective use. Many are saying that those treatments do not whiten as well as the take-home tray systems because they essentially dehydrate the teeth only making them their whitest in the short run, and you still have to use a tray whitening system after the initial treatments to get the best long term results.
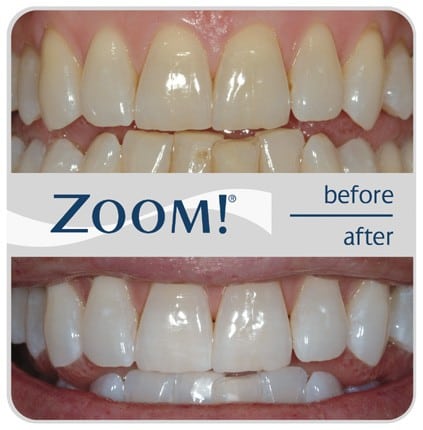
We have the ZOOM (ultraviolet, in office whitening) system at our office, and have actually stopped using it. Not only was it a hassle to use (tying up 2 hours of chair time and one assistant), but the dental assistants don’t like doing it, either. The potential for very sensitive teeth, and gums after the treatment, compounded by unpredictable whitening results, lead me to abandon this type of treatment all together for the more reliable take-home tray systems.
The simplest way to begin whitening your teeth is to get your teeth cleaned. This removes most of the external stains that are accumulated on the outsides of the teeth. This external stain comes from everyday foods and beverages like coffee, tea, soft drinks, blueberry pie, red wines, etc. The actual color of your tooth comes from the internal tooth structure called the dentin. Toothpastes and dental cleanings cannot change this color and can only be changed by bleaching the teeth over a period of time.
A popular, but less effective way to whiten your teeth is though over-the-counter  whitening strips. Huge amounts of advertising dollars have gone into promoting these types of teeth whitening aids. Impulse buying and no need to go to the dental office has boosted sales of these strips around the world. It is great marketing, but a lack luster product at best. I find the product lacking in its ability to whiten the entire tooth structure. After all, it is just a peroxide gum coating a plastic strip that doesn’t exactly fit every little tooth groove and spaces in the mouth. For this reason it has a tendency to whiten the most where it touches the tooth, and not so much between the teeth leaving the teeth with a less than natural look.
whitening strips. Huge amounts of advertising dollars have gone into promoting these types of teeth whitening aids. Impulse buying and no need to go to the dental office has boosted sales of these strips around the world. It is great marketing, but a lack luster product at best. I find the product lacking in its ability to whiten the entire tooth structure. After all, it is just a peroxide gum coating a plastic strip that doesn’t exactly fit every little tooth groove and spaces in the mouth. For this reason it has a tendency to whiten the most where it touches the tooth, and not so much between the teeth leaving the teeth with a less than natural look.
There has got to be at least 20 different whitening gels sold on to the dental profession today. Even more are being pushed by practically every compounding pharmacy in this country. We have found the best products for our patients come from the recommendations of Dr. Gordon J. Christensen. His highly acclaimed Clinicians Report is dentistries version of Consumer Report. It is an unbiased look at top dental products dentists want to use on our patients in our everyday dental practices. Currently we are using Colgate’s Visible White Take-home with custom trays. This product has 9% hydrogen peroxide for whitening. We also are using Platinum Whites with 30% carbamide peroxide and 5% potassium nitrate for tooth sensitivity.
practices. Currently we are using Colgate’s Visible White Take-home with custom trays. This product has 9% hydrogen peroxide for whitening. We also are using Platinum Whites with 30% carbamide peroxide and 5% potassium nitrate for tooth sensitivity.
It is very important that before you spend any money to get your teeth whitened that you get a dental exam and a good professional cleaning first.
Why?
- The dentist can see if you have any decay in your teeth and/or periodontal disease. Decay can begin to hurt and periodontal disease will decrease the effectiveness of the whitening solution/gel.
- A coating of plaque on the teeth will inhibit the peroxides from bleaching the tooth structure. In other words, the teeth must be clean and dry when placing the bleaching product onto the teeth.
- If you have white colored fillings in your front teeth the whitening may make them more obvious. Since your current white filling were matched to your current color tooth, then whitening may make them less attractive. This could cost you more money in the long run by having to replacing your current white fillings with whiter ones to match your whiter teeth.
- If you whiten your teeth before you have white filling done in your front teeth then as the whitening wears off and your teeth fade to their original color (1-2 years) the filling will not match your teeth. This scenario is usually not as much of a problem because white fills blend in better if they are slightly whiter than the tooth structure verses if they were darker.
- Over bleaching can actually cause loss of enamel, make the teeth more brittle, and increase the porosity of enamel making it easier to stain with food and beverages.
- People who have really white teeth have their mouths stared at more than other parts of their body.
It saddens dentists when patients come into their offices and have obvious decay throughout their mouths and are only interested in having their teeth whitened. It just signals to everyone in the dental office that they will need to educate and increase that patient’s dental IQ.
Whitening Physiology
Hydrogen peroxide (H2O2) is a slightly acidic oxidizing agents. When combined with an organic (carbon) source such as a tooth, it will ultimately breakdown everything down to CO2 and water. It does this by breaking down hydrogen peroxide into an acid group (HO2) and oxygen. But we don’t let it react long enough to damage the tooth structure! Instead, it is buffered to a pH of 9-10 to produce an abundance of HO2 that can enter the organic tooth structure (dentin), and altering molecules until they look lighter in color. It is similar to cooking an egg. You don’t say to your spouse, “Honey, would you please bleach me an egg,” yet that is exactly what they are doing.
References:
1. BMC Oral Health. 2011 Feb 23;11(1):6. [Epub ahead of print] Factors influencing patient satisfaction with dental appearance and treatments they desire to improve aesthetics. Tin-Oo MM, Saddki N, Hassan N.
2. Northwest Dent. 2010 Nov-Dec;89(6):19-21, 23-6. Dental whitening–revisiting the myths. Perdigão J. University of Minnesota School of Dentistry, Department of Restorative Sciences, Division of Operative Dentistry, USA. perdi001@umn.edu
3. http://www.amjdent.com/Archive/2007/Sept%202007%20Special%20Issue.pdf
4. J Am Dent Assoc. 1987 Dec;Spec No:44E-52E. Bleaching teeth: new materials–new role. Goldstein RE. Medical College of Georgia, School of Dentistry, Augusta.
5. Compendium. 1990 Feb;11(2):86, 88-90, 92-4. Vital tooth bleaching: the White and Brite technique. Darnell DH, Moore WC.
6. http://jada.ada.org/cgi/content/full/131/10/1478
7. http://www.adha.org/downloads/sup_whitening.pdf
8. Haywood VB, Leach T, Heyman HO, et al.: Nightguard vital bleaching: Effects on enamel surface texture and diffusion. Quintessence International 1990;21:801-806.
9. Quintessence Int. 2009 Apr;40(4):e1-6. Evaluation of 30% carbamide peroxide at-home bleaching gels with and without potassium nitrate–a pilot study. Gallo JR, Burgess JO, Ripps AH, Bell MJ, Mercante DE, Davidson JM. Department of Comprehensive Dentistry and Biomaterials, School of Dentistry, Louisiana State University, New Orleans, LA 70119, USA. jgallo@lsuhsc.edu
10. J Clin Dent. 2005;16(1):17-22. Brushing with a potassium nitrate dentifrice to reduce bleaching sensitivity. Haywood VB, Cordero R, Wright K, Gendreau L, Rupp R, Kotler M, Littlejohn S, Fabyanski J, Smith S. School of Dentistry, Medical College of Georgia, Augusta, GA, USA.

